Science of Saccharin, hooray for science!!!
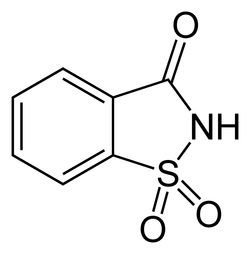
_How to obtain it: Originally take tulane, and then sulfonate it through chlorofluosulfonic acid, then seperate and convert the ortho from the chlorosulfonic acid into sulfonamide by means of ammonia , and then when oxygenated, the substance will crystallize into saccharin.
Molecular Formula: C7 H5 NO3 S
Molar Mass: 183.18 g mol^−1
Visual: White/ Crystally
Mass: 0.828 g/cm3 (but wait, I thought mass was Physics, not Chemistry...)
Melting Point: 228.8-229.7 °C
Boiling Point: 229°C
Solubility in Water: 1 g per 290 mL
Density in natural state: .828 g/cm^3
Reaction Tendencies: Doesn't tend to react with other acids or food. Saccharin soluble in water, but not in acid form. Therefor, the artificial sweetener is typically in the sodium form.
Percent Composition by mass: Carbon49.5%Nitrogen7.65%Sulfur17.5%Hydrogen2.75%Oxygen26.2%
Percent Composition by number: 68.11 %
For more cool sites on different chemical compounds visit http://chemicalcompoundswebproject.weebly.com/
Molecular Formula: C7 H5 NO3 S
Molar Mass: 183.18 g mol^−1
Visual: White/ Crystally
Mass: 0.828 g/cm3 (but wait, I thought mass was Physics, not Chemistry...)
Melting Point: 228.8-229.7 °C
Boiling Point: 229°C
Solubility in Water: 1 g per 290 mL
Density in natural state: .828 g/cm^3
Reaction Tendencies: Doesn't tend to react with other acids or food. Saccharin soluble in water, but not in acid form. Therefor, the artificial sweetener is typically in the sodium form.
Percent Composition by mass: Carbon49.5%Nitrogen7.65%Sulfur17.5%Hydrogen2.75%Oxygen26.2%
Percent Composition by number: 68.11 %
For more cool sites on different chemical compounds visit http://chemicalcompoundswebproject.weebly.com/
